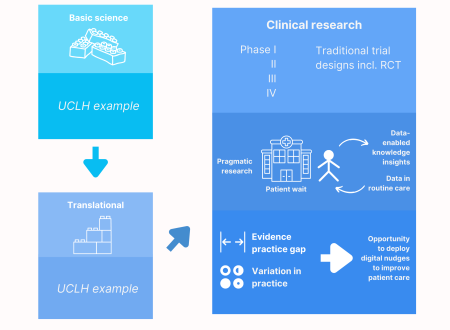
The volume of healthcare data continues to grow, as does the work required to analyse it. However this has yet to produce a significant number of actionable insights that could improve patient outcomes. We do not yet have a strong evidence base for many treatments and interventions given in routine clinical care. Clinical actions are currently driven by expertise instead of evidence from randomised controlled trials (RCTs) that are expensive, labor and time intensive to conduct.
What do we want to do?
Data enabled trials in secondary care take on two major forms:
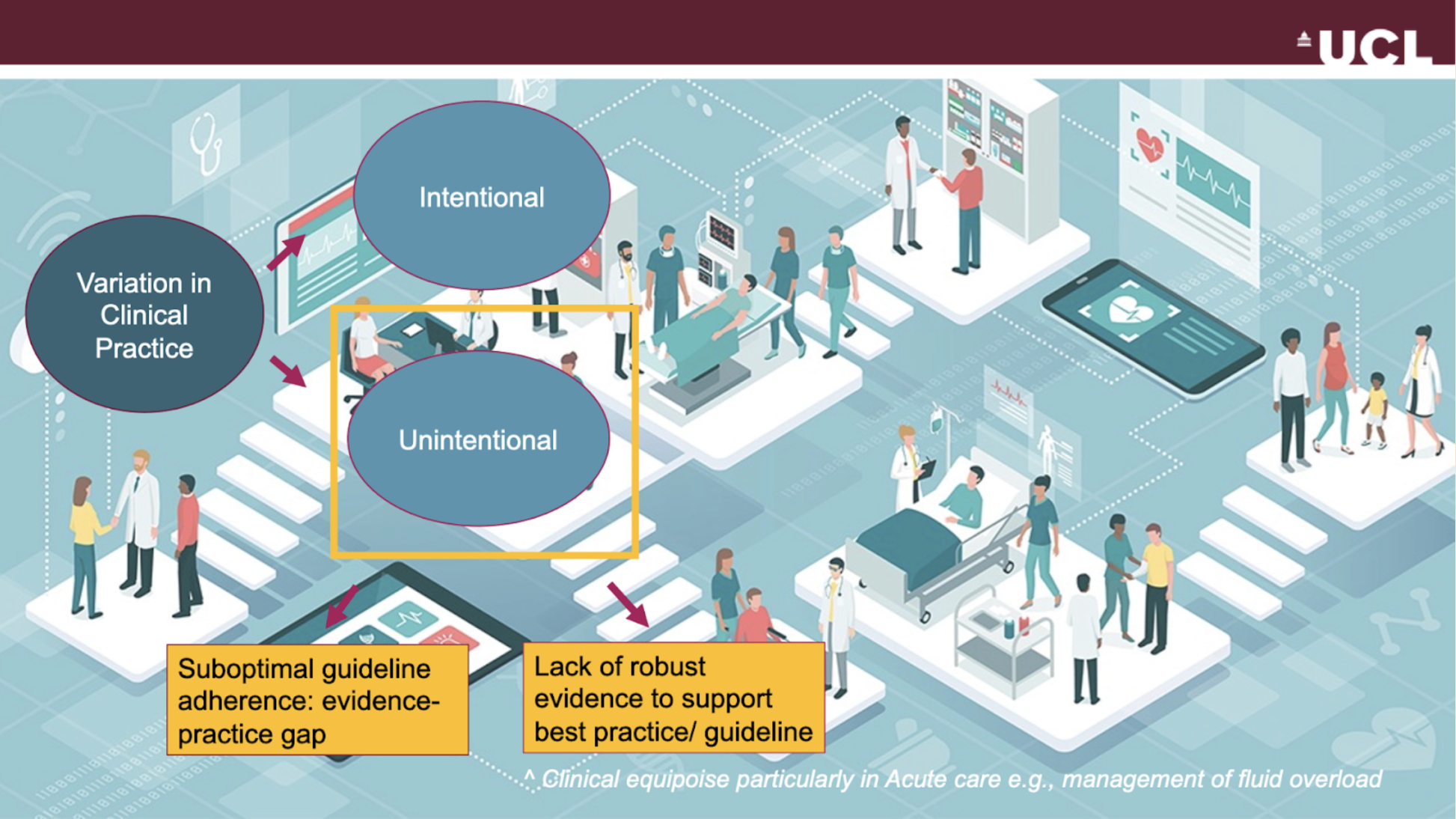
- Observational analyses that can inform future hypotheses for example by spotting where unwarranted variation in practice occurs and identifying whether this is due to an evidence practice gap or a lack of strong evidence in the first place
- Pragmatic research interventions for example nudges as to choice of intervention and option to be included in randomised trial, e.g. nudges which might look like the following:
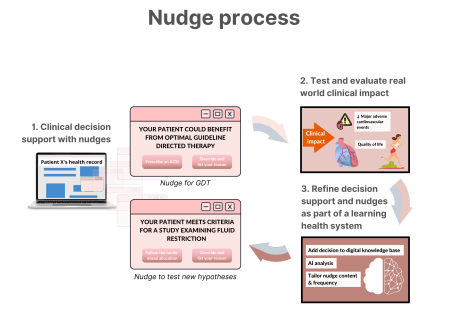
Schematic of a future learning health system with nudge, clinical decision support systems (CDSS), and artificial intelligence-powered analyses forming core parts of the learning cycle.
Nudges within the CDSS can be used to either promote better adherence of guideline-directed therapy or test new hypotheses whenever there is clinical equipoise, identified through variation in clinical practice owing to the lack of robust evidence in that context.
Clinical outcomes could include patient reported outcome measures. The presentation of nudges can be adjusted as new clinical insights are generated either within the local system or externally through data linkage. ACEi, angiotensin-converting enzyme inhibitor; GDT, guideline-directed therapy.
(diagram to the right adapted from The Author(s) 2022. Published by Oxford University Press on behalf of European Society of Cardiology.)