Enhanced Liver Fibrosis test detects, monitors and predicts outcomes of chronic liver disease
The Enhanced Liver Fibrosis (ELF) test, validated at UCLH-BRC, can be used to detect, measure and monitor asymptomatic liver damage. It is the leading prognostic tool for predicting clinical outcomes in chronic liver disease (CLD). With CE mark and FDA approval, ELF is widely used in the NHS.
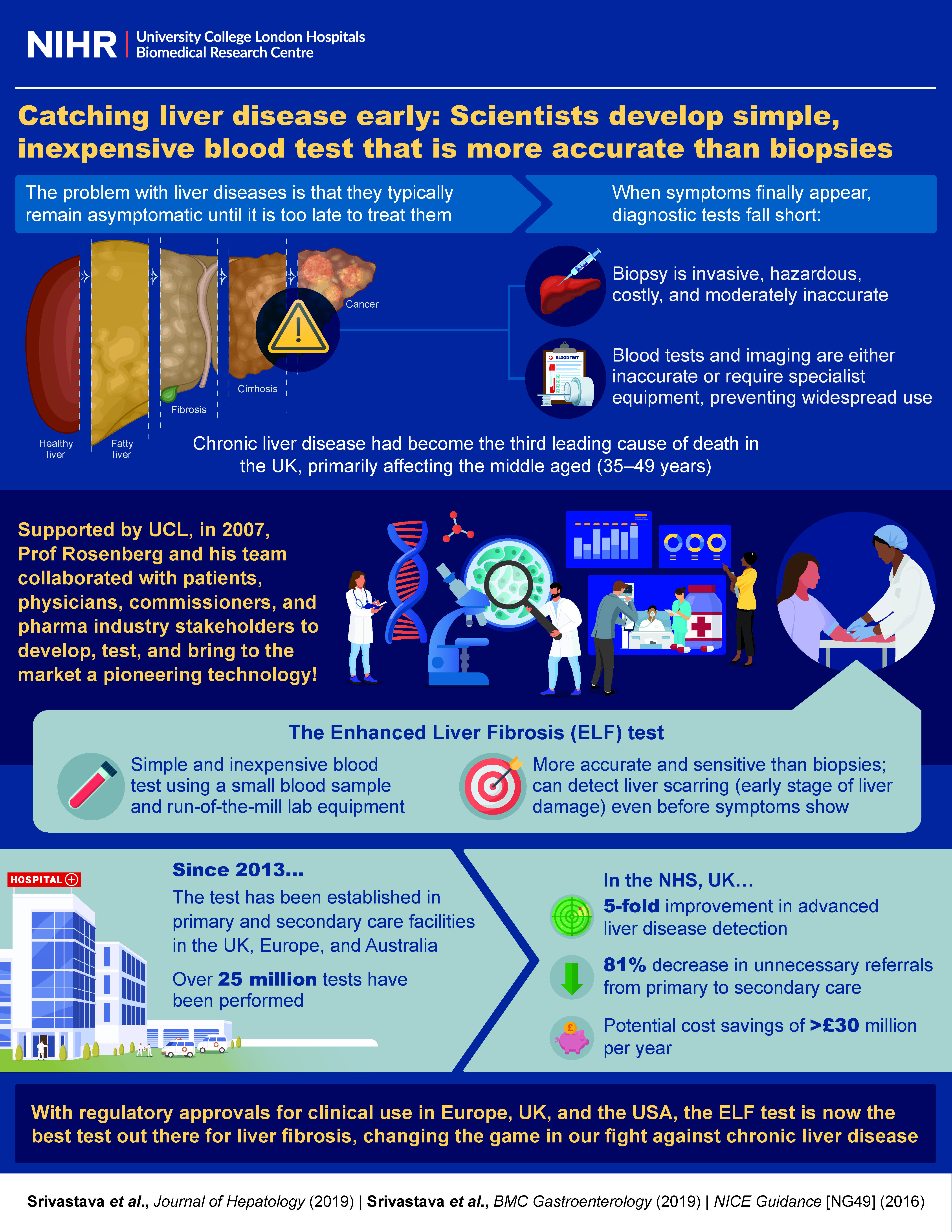
CLD is the third leading cause of death in the UK and the most common cause of death in people aged 35-49. Liver damage (fibrosis) is asymptomatic, with most cases diagnosed when it is too late to treat. Interventions to prevent and reverse serious disease have been hampered by the lack of accurate diagnostic tests. Liver biopsies are painful, hazardous, costly and can be inaccurate. Simple blood tests and imaging are either inaccurate or require specialist equipment, limiting widespread use.
The ELF test emerged from research led by Professor Rosenberg whilst at the University of Southampton (1998), supported by the NIHR Clinical Research Facility. His team identified and developed an accurate, sensitive test for liver fibrosis performed using a small blood sample.
Following the team’s move to UCL (2007) and supported by the UCLH-BRC, Rosenberg led a series of global validation studies. The work involved multiple stakeholders including patients, physicians, commissioners and pharmaceutical companies to optimise implementation and use of the test. ELF outperforms biopsy as a prognostic and monitoring test for CLD and is more sensitive, safer and cheaper.
ELF obtained CE marking in 2011 and was recommended in NICE Guideline NG49 for diagnostic testing for liver disease in 2016(1). It is recommended in British Society for Gastroenterology guidelines and gained FDA approval in 2021.
Following its introduction in NHS clinics, ELF has aided a 5-fold improvement in detection of advanced liver disease and an 81% reduction in unnecessary referrals from primary to secondary care(2) with potential cost savings to the NHS exceeding £30 million per year(3).
Since 2013 over 25 million ELF tests have been performed. The ELF test is established in primary and secondary care pathways for asymptomatic non-alcoholic fatty liver disease and alcohol-related liver disease in the NHS, Europe and Australia. It is widely used in the pharmaceutical industry in drug development to monitor the effectiveness of new medicines.
References:
- NICE Guidance No.49, 2016
- Srivastava A et al. Journal of Hepatol, 2019
Srivastava A et al. BMC Gastroenterol, 2019