THIRST Alert Trial
– a Randomised Controlled Trial within the electronic Health record of an Interruptive alert displaying a fluid Restriction Suggestion in patients with the treatable Trait of congestion
Background
The overall aim of the THIRST trial programme is to determine whether fluid restriction improves clinical outcomes in patients admitted with fluid overload and treated with an intensification of loop diuretic therapy. This is important because there is a lack of robust evidence to support the safety or efficacy of oral fluid restriction in this setting, and because the intervention may be associated with adverse patient experience such as increased thirst.
The need for new evidence on the safety and efficacy of oral fluid restriction remains a gap in the evidence in international guidelines for heart failure. To address this gap in evidence, THIRST will adopt a pragmatic randomised controlled trial (RCT) design embedded in the electronic health record system (EHRS). The intervention is an automatically triggered clinical decision support system (CDSS) alert delivered to the usual care team and treating doctor. THIRST will be delivered in two phases:
(1) THIRST-Alert – the purpose is to investigate the effect of an EHR-embedded randomised CDSS alert suggesting restricted or unrestricted fluid intake on clinical behaviour and on patient oral fluid intake. Outcome measures will include whether the recommendation on oral fluid intake has been actioned by the medical and nursing teams and whether fluid balance measurements have been appropriately documented. The feasibility of performing a large EHR embedded clinical outcomes trial will be evaluated.
(2) THIRST-Outcomes– the clinical outcomes phase will investigate whether fluid restriction has a beneficial impact on length of hospital stay, measures of patient experience, and other relevant clinical safety and efficacy outcomes. Progression to THIRST-Outcomes will be contingent on THIRST-Alert demonstrating that the CDSS alert for restricted or unrestricted fluid intake is effective at modifying oral fluid intake and data quality from routine care is of sufficient quality.
The aim of the THIRST Alert trial is to determine whether restricting fluid intake improves outcomes in patients admitted to hospital having already been treated with diuretics to remove excess water (fluid overload). One of the main causes of fluid overload is heart failure. A lack of strong evidence to support the safety or effectiveness of oral fluid restriction in the acute setting causes significant variation in clinical practice. Some patients are told to restrict fluid and others are not, based primarily on preference of the treating clinician rather than evidence from research.
THIRST Alert is a pragmatic randomised controlled trial (RCT) embedded in the Electronic Health Record System (EHRS). Pragmatic is defined as examining whether interventions work as part of routine NHS care rather than within a formal research setting. Consequently, the THIRST Alert study will only last for 6 months to examine the feasibility of conducting a larger scale study.
The intervention triggering fluid restriction is a EHRS alert to the treating clinician when they prescribe an increased dose of diuretic. The clinician will be asked to consider patients for the study only if there is uncertainty about whether fluid restriction will cause benefit or harm. If appropriate, the patient would be randomised to either restrict oral fluid by 1 litre per day or no restriction.
The study will record the number of patients recruited from routine care, the effect of the alert on clinician behaviour and patients’ subsequent oral fluid intake. The alert only goes to the clinician with no additional burden on patients. The trial requires a waiver of consent; patients will be informed of their participation on discharge from hospital. The trial starts on admission and finishes on discharge from the study site.
As of May 2023, the study has begun recruiting at UCLH. We expect to announce the study results in August 2023.
Why did you choose to study fluid restriction?
Fluid restriction is a common recommendation given to patients. At UCLH, it is given to some but not all people who are admitted with fluid overload. The trade-off for that is considered is potentially increased patient discomfort (and the sensation of thirst) for improved treatment of their symptoms of fluid overload.
There is surprisingly no robust evidence to support its use in the acute hospital setting, and so whether patients receive fluid restriction or not is down to the preference of the treating team rather data generated from scientific research studies.
Fluid restriction is a behaviour rather than a medicine or intervention - the patient remains ultimately in charge of how much they drink. This makes THIRST a particularly low-risk study and this is reflected in the proportionate consent used (verbal opt-out).
What do you mean by 'pragmatic randomised controlled trial (RCT) design embedded in the electronic health record system (EHRS)?
Unlike a traditional RCT, THIRST has the following unique features: (1) There are no additional research resources required such as having a dedicated research team or nurses at the study site. (2) There will be no additional burden to patients as there is no additional blood tests or follow up required - the trial concludes upon patient discharge. (3) The identification of eligible patients and the randomisation process is done by the EHR in an automated fashion (4) There is no additional resource required to deliver the intervention or to measure the trial outcomes - we are evaluating whether this can be done by the routine care team.
As a result of our design choices, THIRST Alert is a highly pragmatic trial that could be generalisable to many hospital settings.
Study team:
- Dr Yang Chen, Principal Investigator and Clinical Research Fellow, yang.chen@nhs.net
- Dr Tom Lumbers, Chief Investigator and Consultant Cardiologist, tom.lumbers@nhs.net
- Cardiology Department at UCLH: +44 (0) 20 3447 8066
References
The PROSPECTOR Feasibility Study
Point of Care RandOmisation Systems for Performing Embedded Comparative Effectiveness Trials of Routine Treatments
This study has finished and results will be updated soon.
Every day, doctors and nurses make hundreds of decisions about how to administer treatments that are considered part of routine care. How much oxygen to administer, what type of intravenous fluid to use, whether to stop antibiotics or continue another day etc. Some of these treatment decisions are supported by good quality evidence from clinical trials, but many remain “evidence-light”.
When a treatment lacks evidence, clinicians base their decisions on a combination of experience and basic scientific knowledge - the lowest levels of evidence based practice. However, they are also influenced by heuristics, and biases which mean that patients can end up receiving varying care depending on who looks after them.
At best, variations in care are intentional, resulting in treatment approaches which are personalised to the patient. At worst, variation may be unwarranted. Ultimately, variations result in improved patient outcomes, worse care, or make no difference at all (indicating the treatment might be ineffective).
Currently we are not able to learn from routine clinical decision making and unwarranted variation persists. We need to be able to learn from every clinical decision which lacks evidence, to improve the care for future patients.
We propose to utilise the uncertainty clinicians experience when administering treatments which lack evidence. Where the clinician has genuine uncertainty about the whether a treatment is good or bad for the individual patient, they are offered the opportunity to follow a randomised treatment suggestion within a clinical trial. This randomisation is flexible, if the clinician knows better than the study, they may follow their treatment preference.
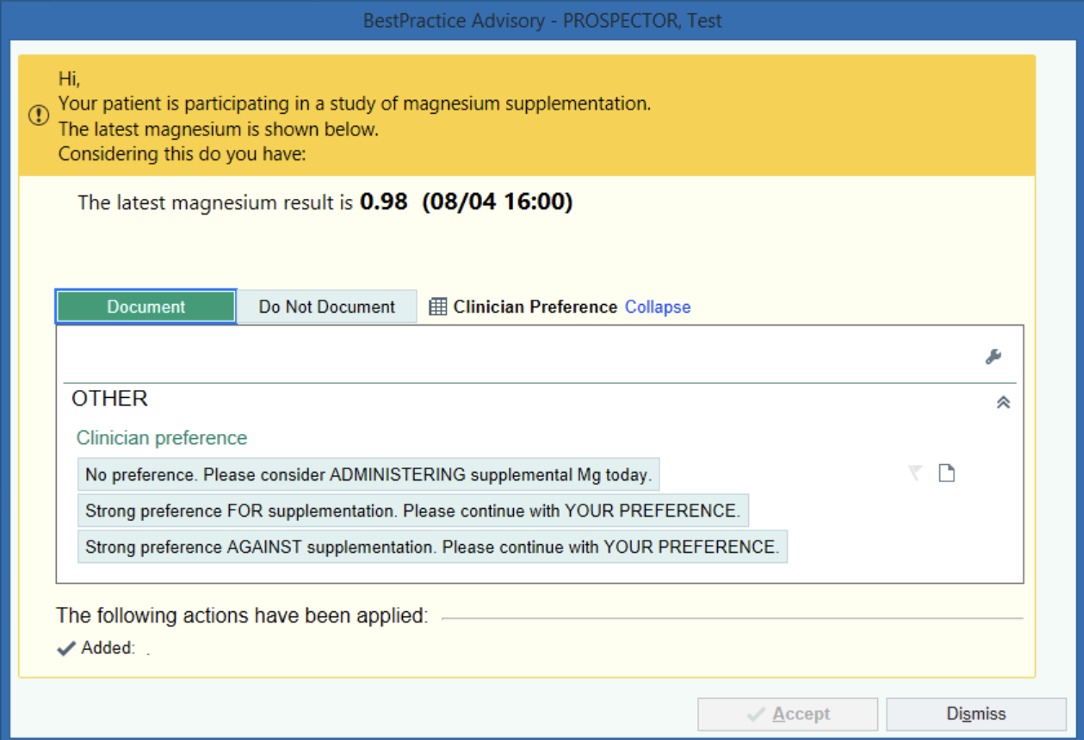
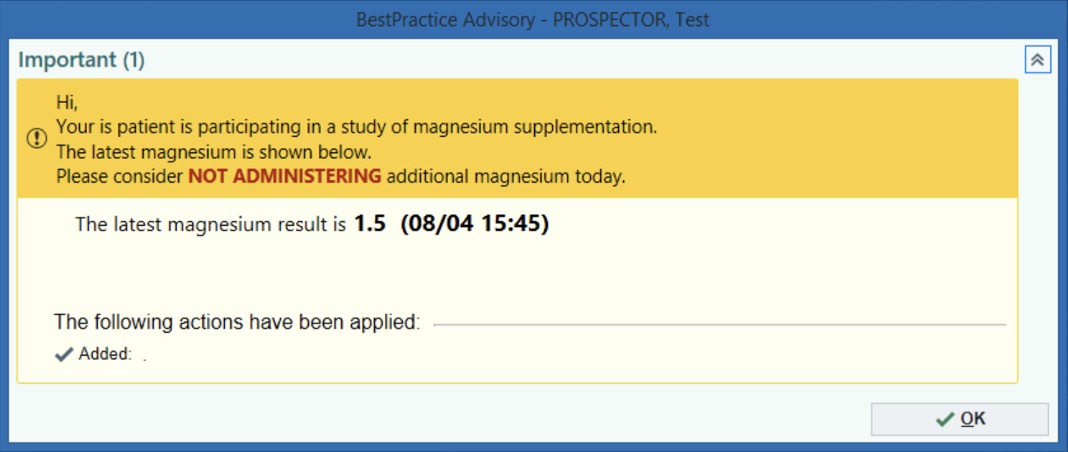
Using existing Electronic Health Record Systems, clinical trials of routine treatments may be integrated within clinical workflows. Patient screening and data collection are automated, and flexible randomisation is offered close to the point of clinical decision, via modified clinical decision support tools.
The PROSPECTOR feasibility study will compare two designs of electronic point of care randomisation prompt to see what is acceptable to clinicians. It also includes a program of qualitative work to explore patient and public attitudes towards consenting to participate in clinically-integrated trials, of naturally varying, low risk routine treatments.
For further study information: https://clinicaltrials.gov/ct2/show/NCT05149820
To view study publications: Publications and impact
For further detail on aspects of the study design, including information on the concepts underlying clinically integrated randomised trials, point of care trials, and preference study designs [click here].
For further detail on development and design of both Nudge and Preference ePOCR prompts [click here].
For further detail on our program of work exploring opt-out consent for trials within Learning Health Systems [click here].
If you would like to contact a member of the study team please see here: Contact us.
Links:
1. Publications list:
- Nil to date….
- Under review:
- BMJ Open Prospector study protocol
- JBI Trial Design Review
- Nat Sci Rep Mg AF IV study
2. Clinically-integrated randomised trials
- Short descriptions of each concept, with text mainly lifted from narrative review
- Some simple diagrams illustrating each concept
3. ePOCR Prompt Design
Technical elements of design
4. Ethics of Learning Health Systems
- Short descriptions of some of the main ethical issues, consent issues
- Links to review work
- Links to opt out consent section
5. Opt out consent
- Short public friendly description
- Embedded PPI video